If the caesium element is irradiated with a wavelength 500 nm, calculate the kinetic energy and the velocity of the ejected photoelectron. the threshold wavelength and the threshold frequency of the radiation. That is equivalent to losing or gaining no more than one second in the age of the universe. The work function for caesium atom is 1.9 eV. This can also be achieved for atoms trapped by intersecting light beams.Īt NPL, optical clocks are being developed which target accuracies of around 100 times higher than the best current microwave frequency atomic clocks. In this way, certain optical absorptions in the ion can exhibit a very pure frequency. Ions are charged atoms which can be trapped almost indefinitely by electromagnetic fields, and cooled by laser beam close to absolute zero. The Global Positioning System (GPS) satellites broadcast timing signals from onboard atomic clocks, which enable land vehicles, shipping and aircraft to know their location within a few metres.Ĭlocks for the 21st century based on single cold trapped ions or collections of atoms are being developed. Time measurement has become a basic part of everyday life and accuracies of the nearest minute or a few seconds are usually good enough for most human activities, but highly accurate timing plays a vital role in many other aspects of the modern world. 
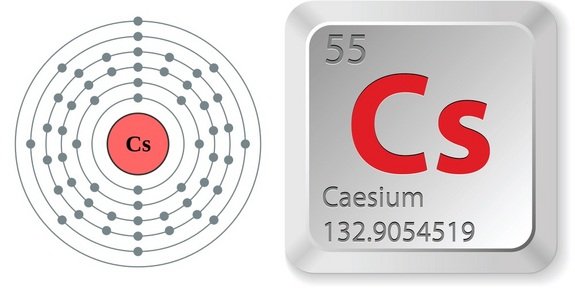
It is a soft, silvery-golden alkali metal with a melting point of 28.5 C (83.3 F). By measuring the frequency of the electromagnetic radiation, like counting the number of pendulum swings, we can measure the passage of time. Caesium is a chemical element with the symbol Cs and atomic number 55. This energy is absorbed or released in the form of electromagnetic radiation, the frequency of which depends on the difference in energy between the two levels. The orbits correspond to energy levels, and electrons can only move between levels when they absorb or release just the right amount of energy. We report on reflection spectra of Caesium atoms in close vicinity of a nanostructured metallic meta-surface. The atom can be pictured as a mini solar system, with the heavy nucleus at the centre surrounded by electrons in a variety of different orbits. However, the quartz and atomic clocks invented during the 1930s and 1950s were even better timekeepers, and showed that the Earth does not rotate steadily but "wobbles". Modern atomic clocks can be as much as a million times more accurate than this, and underpin satellite technology, like GPS or the internet.įor thousands of years the Earth's rotation was our most stable timekeeper. This clock was so accurate that it would only gain or lose one second in three hundred years. NPL developed the first operational caesium-beam atomic clock in 1955. However this period of rotation is not regular enough to serve as a definition for modern-day applications.Ītomic clocks, which keep time using transition energies in atoms, revolutionised timekeeping. These methods of timekeeping were based on the daily rotation of the Earth around its own axis. As well as enabling us to tell the time of the day, accurate timekeeping is key to satellite navigation systems, underpins the functioning of the internet and facilitates timestamping for transactions in financial trading.Īncient civilisations used sundials and obelisks to tell the time, which was not very precise, and was restricted in cloudy weather or at night. cesium (Cs), also spelled caesium, chemical element of Group 1 (also called Group Ia) of the periodic table, the alkali metal group, and the first element to be discovered spectroscopically (1860), by German scientists Robert Bunsen and Gustav Kirchhoff, who named it for the unique blue lines of its spectrum (Latin caesius, sky-blue). Phenom., 1980, 21, 275.The second is used to measure time. Mårtensson, "Core-Level Binding Energies in Metals," J. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. If the caesium element is irradiated with a wavelength of 500 nm, calculate the kinetic energy and the velocity of the photoelectron. Calculate (a) the threshold wavelength and (b) the threshold frequency of the radiation. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). Click hereto get an answer to your question The work function for the caesium atom is 1.9 eV. The data are adapted from references 1-3. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. 
All values of electron binding energies are given in eV. 1967, 47, 1300.Įlectron binding energies Electron binding energies for caesium. These effective nuclear charges, Z eff, are adapted from the following references:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
